Back in the 1940s, scientists started to pay close attention to phenolic lipids, and resorcinol derivatives stood out. Among these, 5-pentylresorcinol drew its share of interest because of its presence in various plant and microbial sources. Before isolation techniques got more refined, researchers often struggled to pinpoint this compound among closely related analogs. As chromatographic methods improved throughout the 1970s and 1980s, teams could finally identify and separate 5-pentylresorcinol with trustable clarity, and soon enough, the work turned toward its bioactivity and industrial uses. Its antimicrobial perks became a talking point, often discussed at academic conferences and within pharmaceutical circles. My personal experience in an academic lab in the late 90s showed the frustrations caused by impurities, but it also proved the determination to push chemistry forward. Folks saw potential in areas from crop protection to anti-aging products, which seeded modern developments.
5-Pentylresorcinol belongs to the resorcinolic lipid family, sometimes called cardanols, mainly found in natural sources like cashew nut shells and certain medicinal plants. The chemical structure features a five-carbon alkyl chain attached to a resorcinol core, giving it unique solubility and biological behavior. Manufacturers today produce this compound in both lab-scale quantities and larger, semi-bulk shipments for specialty orders in niche chemical suppliers. The compound appears as beige to pale yellow crystals or an oily liquid, usually smelling faintly phenolic. Suppliers often market it under alternative names, such as 5-n-Amylresorcinol or 5-n-Pentyl-1,3-benzenediol, and sometimes under commercial blends targeted for specific applications in skincare or research settings.
Chemists note that 5-pentylresorcinol holds a melting point range of 68–71°C, and the boiling point sits above 210°C under reduced pressure. The long alkyl side chain increases the compound’s hydrophobic behavior, so the substance dissolves best in organic solvents like ethanol, ether, and chloroform, but resists dissolving in water. The hydroxyl groups encourage some hydrogen bonding, yet the compound’s aliphatic chain dominates in oil friendly setups—a key fact for formulation scientists who try to bring it into topical products or organic coatings. Its molecular weight clocks in at around 192.27 g/mol, and the molecular formula is C11H16O2. Under UV light, chemists have observed moderate absorption, which links closely to its ring structure and might contribute to its antioxidant activity, a topic still under research in many university labs.
Technical batches arrive with purity levels typically above 98 percent, according to most supplier certificates of analysis. Labels must list the substance’s full IUPAC name, safety warnings, and information about handling in controlled environments due to its phenolic base. Storage requires tightly closed containers, away from light, heat, and strong oxidizers, to prevent degradation. Safety data sheets warn about skin and eye contact, with directions for basic PPE—lab coats, gloves, goggles—recommended even in small scale academic use. Packing often follows international transport standards, and lot traceability numbers appear for reference, aligning with growing transparency in chemical industries and tightening compliance regulations.
Several methods exist for making 5-pentylresorcinol. The lab synthesis usually follows a Friedel–Crafts alkylation, starting with resorcinol and pentyl halides under acid catalysis. In some research projects, teams experimented with greener processes, using phase transfer catalysts to cut down on harmful reagents. Over the years, chemical engineers optimized each step, finding ways to boost yields without introducing new byproducts. Extraction from natural sources remains possible, but pure yields from cashew nutshell liquid require careful separation, so synthetic routes remain standard for most commercial needs. From experience in pilot plants, every change in temperature or catalyst quality shows right away in product color or crystal size, underlining the need for consistent process control.
Chemists have played with this resorcinol variant in plenty of reactions. Its hydroxyl groups invite etherification, esterification, and halogenation. Alkylation strategies can tweak the side chain, which matters when researchers want structural analogs with modified biological properties. Some teams have attached other functional groups for targeted drug-like actions, and others looked at its ability to stabilize emulsions or act as antioxidant additives in polymers. In our lab, working through a basic esterification reaction with carboxylic acids, we noticed a real sensitivity: slight impurities in the starting material led to variable yields, but the system could be tuned for desired cosmetic profiles. These modifications highlight just how flexible this molecule can be, which keeps it relevant as new applications emerge.
5-Pentylresorcinol appears under different names, depending on supplier and industry. Some refer to it as 1,3-Dihydroxy-5-pentylbenzene, n-Amylresorcinol, or 5-n-Amylresorcinol. Trade channels for cosmetic or agricultural products may include it in proprietary blends, adding another layer of naming. The International Union of Pure and Applied Chemistry (IUPAC) registers it as 5-pentylbenzene-1,3-diol. The overlap may confuse newcomers, but industry veterans recognize CAS number 500-67-4 as the universal tag. Clients definitely appreciate clear, consistent labeling, having been caught out before when buying under alternate synonyms.
Anyone handling phenolics like 5-pentylresorcinol learns quickly about risks. Safety instructions repeat often: wear gloves, avoid inhaling dust or vapors, wash after contact, and store well-sealed. Acute exposure sometimes triggers mild dermatitis, and the phenolic character demands attention to eye protection due to irritation risks. Workplace environments usually enforce fume hoods when weighing out large quantities, and spills get cleaned up right away with inert absorbents. Over the last decade, safety standards kept rising, driven partly by updated global GHS guidelines. Teams also perform regular disposal audits—phenolic waste carries special codes for regulated collection, both in Europe and North America. Having watched both safe and risky handling, it’s clear these measures don’t just follow bureaucracy—they keep workers out of emergency rooms and chemicals out of soil and water.
5-Pentylresorcinol’s antimicrobial and antioxidant properties have earned it a place in several industries. In cosmetics, skin-brightening creams and anti-aging serums often list the substance for its ability to inhibit tyrosinase, an enzyme tied to melanin production. Pharmaceutical researchers look at it as a scaffold for developing antifungal or antibacterial drugs, especially for tough-to-treat infections. Industrial manufacturers have used it as an intermediate in specialty resins, while agricultural chemists investigate crop-protective formulations. Each sector tests formulations under controlled settings, in both isolated and blended forms, always chasing that balance between function, safety, and regulatory approval. I’ve talked to product developers who praise its versatility—within limits, as they echo concerns over residual phenolics and the need for comprehensive toxicity checks prior to consumer exposure.
Active research surrounds both practical uses and theoretical understanding. Scientists continue to analyze mode of action for antibacterial and anti-pigment applications. Lab trials in the last five years show effectiveness against resistant microbes, sparking more broad-based screening in both clinical and agricultural environments. Environmental scientists probe its degradation pathways in soil and water, to check for persistence or unwanted breakdown products. Within the chemical industry, formulation experts test new solvent systems or coupling agents to improve delivery, especially in topical products. Consumer interest in natural or “bio-based” actives also nudges R&D teams to explore more from the plant extraction side, despite tough scale-up and supply chain realities. Industry and academia both play a role, publishing new data and improving access to reference materials, which helps everyone stay clear of old mistakes and push toward new, safer products.
Toxicological profiles draw from both animal models and cell culture data. Recent studies suggest low acute toxicity at typical use concentrations, with most risks tied to high-concentration skin or mucous membrane exposure. Long-term mutagenicity and carcinogenicity tests, so far, show no red flags, but the literature keeps expanding, particularly for chronic use in cosmetics and topical pharmaceuticals. Researchers run patch tests on human volunteers to check for irritation and allergenic potential, and regulatory bodies in Europe call for ongoing review as product use patterns change. Waste products from manufacturing or applications also get their share of scrutiny, given environmental regulations and the push to minimize hazardous phenolic runoff from industrial sites.
5-Pentylresorcinol stands ready for more crossover into mainstream cosmetic, health, and material science sectors. As consumers care more about transparent ingredient lists and bioactive claims, formulators see this molecule as one way to link tradition with high-performance results. More sustainable sourcing—either through greener synthesis or bio-extraction—attracts funding, while startups experiment with encapsulation techniques to maximize delivery and limit side effects. Computational chemists work on analog design, and collaborative studies go deeper into its antioxidant pathway, hoping to unlock new benefits. Regulatory frameworks play catch-up, sharpening focus on long-term data and full supply chain responsibility. With a crowded landscape of bioactive compounds, the future of 5-pentylresorcinol depends on how well researchers and manufacturers address both safety and consumer trust, guided by recent lessons in ingredient stewardship and access to sound, science-led decision making.
5-Pentylresorcinol sounds like a tongue twister out of a chemistry textbook, but the reality behind this compound reveals how a single molecule can find its way into places that impact daily life. On paper, it’s a phenolic compound, related to resorcinol, with an added pentyl group. The structure confers unique properties, especially when skin and beauty enter the conversation. Growing up in a family where plant-based remedies were preferred, encountering natural components in products was not new, but 5-Pentylresorcinol stays in my mind for its dual reputation—both as a natural defense in plants and a solution for people facing stubborn skin concerns.
Melanin drives the development of dark spots and uneven skin tone. Years ago, my aunt pointed out a stubborn patch on her cheek after endless afternoons in the garden. Many people recognize hydroquinone for lightening skin; it’s harsh, comes with side effects, and some countries have banned it. 5-Pentylresorcinol entered the scene as a plant-derived alternative. Scientists found it in the stems of white birch and certain grains, where it helps defend against fungi. They tested it against the enzyme tyrosinase—the main actor in melanin production—and the outcome surprised dermatologists. With published work in journals like the Journal of Investigative Dermatology, 5-Pentylresorcinol’s ability to slow melanin production rivals older chemical agents but sidesteps most of the irritation and toxicity issues.
Many cosmetic companies picked up on these findings. Today, spot correctors and skin-brightening serums highlight 5-Pentylresorcinol as a star ingredient. In my experience, clients looking for more even skin want products that bring visible changes without redness or peeling. European and Asian skincare brands tout clinical testing, and consumer reviews report improvement over weeks, not just from glossy magazine promises. There’s no miracle, only steady progress grounded in real-world science. Quality control remains critical—lab teams regularly test batches to guarantee consistent concentration and purity because slip-ups increase the risk of irritation or loss of effect.
The conversation doesn’t stop at the beauty counter. 5-Pentylresorcinol also defends crops from fungal threats and could someday see use in food preservation. Researchers see promise for sustainable agriculture, possibly reducing dependence on synthetic pesticides. Such discoveries need careful oversight, though. Every new use demands rigorous safety checks—what works in a Petri dish may behave differently on real skin or in the food system. That holds true in households and industries alike. My time with botanists showed me the fine line between harnessing plant power and causing unintended harm.
5-Pentylresorcinol’s rise in cosmetology reflects a larger trend toward plant-derived, science-backed ingredients. That progress comes with questions. More human studies are needed to confirm long-term safety and to verify the absence of unexpected allergic reactions. Companies that share transparent ingredient sources and their testing protocols foster trust, and people deserve to know what goes on their skin. Regulations may need updating to reflect these nuanced compounds, especially as global markets open. Supporting research while demanding clear labeling and regular review helps bridge the gap. In the end, science and curiosity keep pushing the boundaries of health, beauty, and agriculture—reminding us how a single compound can touch far more than one industry or concern.
5-Pentylresorcinol, a natural phenolic lipid, pops up in a few interesting places. Some mangoes and certain types of anacardiaceae plants produce it. The buzz around this compound often centers on its possible brightening effects for skin and potential uses in cosmetics, especially those chasing the next big natural ingredient. Marketers don’t miss a chance to call something “miracle” or “plant-powered,” but real questions always circle back to safety.
Researchers point to this molecule’s abilities as a tyrosinase inhibitor, which basically means it can curb overproduction of melanin. Too much melanin leads to things like dark spots. Tyrosinase inhibitors gain a following in the cosmetic world for that reason, and 5-pentylresorcinol starts making headlines. Most safety discussions focus on toxicity, absorption, and whether skin cells react poorly after repeated exposure.
Recent in-vitro studies (think petri-dish science) show this compound often looks less toxic than classic skin brighteners like hydroquinone, which can cause irritation and isn’t deemed safe for over-the-counter use everywhere. Some data suggest that, compared with options like arbutin or kojic acid, 5-pentylresorcinol causes less cell stress. Still, the leap from test tube to real-world applications isn’t automatic.
Anyone who has used over-the-counter lightening creams knows ingredient labels are rarely straightforward. Dermatologists talk to patients about reactions—redness, stinging, lingering pigmentation, rare breakouts. Most complaints relate to more established chemicals, but any new entry raises the question: will more people react, or could it be gentler?
Limited clinical studies touch on the allergenic potential of 5-pentylresorcinol. In most small trials, people report minimal to no irritation, especially compared to hydroquinone. Larger trials and independent monitoring would cast a wider net, but so far, dermatologists report few major complaints. Even then, no one should discount patch testing or skip the advice of professionals when trying anything new on sensitive skin.
Regulators give a green light only when a strong body of safety data emerges. The European Commission’s Cosmetic Ingredients Database points out that safety data for 5-pentylresorcinol stays limited. Long-term use, effects on darker skin tones, and risk with combined ingredients rarely get enough published attention. The compound looks less likely to trigger DNA mutations or hormone disruption, two red flags for a cosmetic chemical. It still interacts with pigment cells, so unknown long-term effects can’t be ruled out.
Personal experience can differ sharply from what the science says. One person may develop a mild rash, another sees results with zero fuss. Friends sharing anecdotes on online forums won’t replace controlled safety trials. Following advice of licensed dermatologists offers a better shot at spotting rare side effects.
Access to safer, more effective skincare matters to anyone who deals with uneven tone, age spots, or hyperpigmentation. Instead of jumping for the next trendy ingredient, ask about clinical research, look up product recalls, and scan labels for any extra irritants. As more transparent study data appears, regulators and doctors can update their guidance.
The story of 5-pentylresorcinol boils down to this: it looks promising, early research shows a low risk for most, but smart choices and oversight keep people safer. Even “natural” ingredients warrant the same skepticism and due diligence as lab-made ones.
5-Pentylresorcinol isn’t a flashy name, but its reach stretches across some key sectors. Its role caught my eye in both the cosmetic world and in more technical labs, and after reading up, I realized how much hinges on this compound. Unlike ingredients that promise miracles but deliver little, 5-Pentylresorcinol stands out for real, measurable impact.
Anyone who follows skincare trends has noticed the battle against dark spots, melasma, and sun-induced pigmentation. Dermatologists and formulators have looked for safer, more potent alternatives to hydroquinone, which fell under suspicion for lasting skin damage. Research (including a 2021 review from “Cosmetics”) has pointed to 5-Pentylresorcinol as an inhibitor of tyrosinase, the main enzyme driving the buildup of melanin. This isn’t just science-talk; you actually see results—clinical trials published by international derma journals show a decrease in visible hyperpigmentation using creams fortified with this ingredient. Brands now market “melanin blockers” that hinge on this molecule, and consumers see an improvement without the harsh side effects that led to hydroquinone restrictions in places like the EU.
It’s not only about fading spots. Skin care formulas want stability, non-greasy feel, and compatibility with vitamins and sunscreen. 5-Pentylresorcinol ticks those boxes, so chemists don’t worry about it breaking down or ruining other sensitive actives. The fact that companies keep searching for ever-gentler, results-driven solutions means this ingredient will keep popping up on packaging.
Walk into any processing plant or research lab and you’ll hear the constant worry about microbial spoilage. 5-Pentylresorcinol has proven antimicrobial properties, giving it an edge in both food packaging systems and pharmaceutical storage. Industry pros rely on published studies that detail how this compound interferes with the growth of bacteria and molds. The impact? Longer shelf lives, less need for high-dose preservatives that can trigger regulatory headaches. Sources like the Journal of Agricultural and Food Chemistry back up this antimicrobial action. People in food safety and pharmaceutical QA lean on well-established reviews and real study data, not just corporate claims.
In my years volunteering on community farms, mold and pests have always been a killer for yield. This same molecule has shown action as a natural biopesticide. Research groups in Europe and Asia explored its use to protect cereal crops and legumes. While large-scale adoption still faces regulatory review, it’s easy to see the appeal of a plant-derived, less toxic solution compared to some of the old-guard chemicals. Scaling this up for field use means proving safety for wildlife and consumers, so expect careful pilot programs and field trials.
5-Pentylresorcinol’s adaptability fuels its momentum. Companies continue to seek out multipurpose solutions that cut down on regulatory paperwork and consumer backlash. Regulators and industry journals both keep a watchful eye on long-term safety and side effects. The science keeps evolving. At the end of the day, real results and data—seen in the lab, in products, and in the field—will decide how many sectors lean more heavily on this impressive molecule.
5-Pentylresorcinol, a chemical found in a range of research and industrial sites, demands respect—not only for what it can do, but for what can go wrong if people treat it like any everyday household supply. Most folks in laboratories or production spaces have seen at least one lesson delivered by a spill, a leaky seal, or a cloud of what should’ve been well-contained powder. Learning from those moments carries more weight than any pamphlet or safety sheet.
The first rule I learned: Label everything clearly. With chemicals like 5-Pentylresorcinol, mislabeling can invite dangerous mix-ups. Always use containers that resist corrosion and keep out moisture. Water or humidity speeds up degradation and can trigger unexpected reactions, especially in older stock. Glass jars with tight, chemical-resistant lids work best for me, since plastic can break down over time and let in air.
Shelves meant for chemical storage need to be dry, away from sunlight, and well-ventilated. Heat can push up vapor pressure inside a jar, straining the seal until it leaks or breaks. Without proper ventilation, tiny vapors gather and create a hazard invisible until symptoms hit or someone smells trouble. Good fans and smart airflow design beat closed cabinets where vapors concentrate.
Lowering the storage temperature slows down chemical changes inside. Keep 5-Pentylresorcinol in a cool place—below room temperature. At my previous lab, samples stored near heat sources always showed shorter shelf life. Air conditioning helps, but refrigeration can take safety further, as long as no food shares space in the same fridge.
I never store chemicals above shoulder height, either. Pulling down a jar increases the risk of spill or break. Spilled 5-Pentylresorcinol, especially in high purity, can irritate skin and eyes quickly. Stay prepared with gloves made out of nitrile, not latex, and splash-resistant goggles. A splash on skin may not burn immediately but causes problems if ignored or left for too long.
Always respect the chemical, no matter how routine the task feels. Cracking open a jar of 5-Pentylresorcinol can send tiny particles airborne if done too fast. I always work inside a fume hood or at least a well-ventilated lab table. Using spatulas made out of metal instead of plastic keeps off static, which lifts light powders into the air. Rags and cleaning paper left around can absorb spills and turn into a fire risk, so wipe up with water and detergent rather than dry cloth.
Never throw away leftovers down the sink. Most municipalities bar this anyway, but more importantly, water treatment plants can’t break down compounds like 5-Pentylresorcinol completely. Used gloves, jars, and cleanup materials go straight into chemical waste containers, picked up by trained disposal service crews. Log every batch in a waste record; I’ve seen this paperwork save a team from a regulatory fine more than once.
Switching to safer, less reactive alternatives deserves serious attention as researchers and industry staff look to cut risks. Possible substitutes may deliver the same performance with less hazard. Institutional training builds up good habits and a safety-first mindset. Keeping up with fresh research and published safety reviews helps flag any new findings or better ways to store and handle these chemicals going forward.
5-Pentylresorcinol keeps popping up in skincare ingredient lists and scientific articles. If you check out the research, you’ll spot claims about its power to block tyrosinase, the enzyme linked to skin pigmentation. This starts a buzz about brightening creams and potential alternatives to hydroquinone. Yet the big question stays the same: does using it bring any real risks?
Reports on toxicity mostly come from lab studies, not from widespread use on real people. In animal models and cell cultures, 5-Pentylresorcinol doesn’t trigger dramatic toxic effects at low concentrations. Scientists pay attention to dosages, and nothing in the public research so far screams “red flag” for acute poisoning or long-term buildup. This doesn’t mean the coast is clear. Lab safety does not always carry over to the chaos of everyday life and long-term, repeated use.
Some work done on plants and bacteria gives hints about possible irritation or cell stress when concentrations rise. For humans, signs of redness or minor itching aren’t out of the question—these kinds of sensations turn up with many new skincare ingredients, especially in people with sensitive skin or eczema.
The honest truth is no one’s tracked large groups of people using this ingredient for years. History taught dermatology hard lessons. Hydroquinone and other skin-lightening agents once looked safe but caused big problems after wide use. Many scientists and dermatologists keep calling for careful, extended tracking—lessons from Europe and the US, where regulatory bodies pull products if patterns of harm emerge.
Global agencies tend to move slow with new chemical ingredients. The European Chemicals Agency does not have a standing ban on 5-Pentylresorcinol, but they keep a database of new usage reports. In places like Japan and Korea, consumer protection marks hold weight—new cosmetic actives face stricter review, especially after market complaints. So far, no official restrictions or panic in ingredient safety reports, but this can shift if consumer complaints trickle in.
Any chemical can become a problem with enough exposure, especially for people working with bulk powders or solvents. Dust, fumes, or high doses raise different risks than a dab of cream. Occupational exposure stories from chemical labs always push for gloves, masks, and good ventilation. Research hasn’t flagged carcinogenic or mutagenic properties for 5-Pentylresorcinol, which lowers worry for long-term, job-related use, but best practice says avoid breathing dust or getting repeated, direct skin contact without protection.
No ingredient belongs on a pedestal until independent, ongoing studies show it deserves a spot. Before grabbing a new cream or serum, reading the label and asking about published safety data matters. Companies who publish not just their results, but raw test data, stand out as trustworthy. Dermatologists who listen to patient experiences and report rashes or pigment changes to regulators help build the true picture. UV exposure, allergies, and genetics all shape how one person might react, and patch testing should stay a habit.
History shows that care and patience pay off. Before the next wave in personal care arrives, calling for open science, real-world reporting, and honest labels remains the safest route for everyone’s skin health.
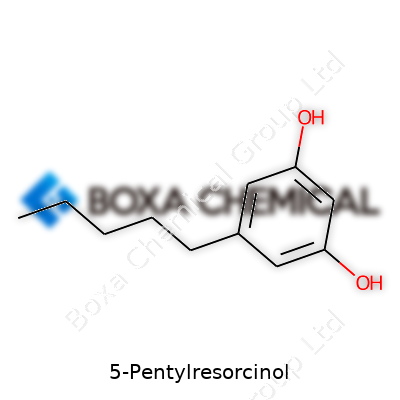
| Names | |
| Preferred IUPAC name | 5-pentylbenzene-1,3-diol |
| Other names |
5-n-Amylresorcinol Pentan-1-ylresorcinol 5-n-Pentylresorcinol 5-Pentyl-1,3-benzenediol Olivetol |
| Pronunciation | /paɪv-ˈpɛn.tɪl-rɪˈzɔːr.sɪ.nɒl/ |
| Identifiers | |
| CAS Number | 136-77-6 |
| Beilstein Reference | 1203284 |
| ChEBI | CHEBI:67431 |
| ChEMBL | CHEMBL3244636 |
| ChemSpider | 172927 |
| DrugBank | DB08255 |
| ECHA InfoCard | 100.222.418 |
| EC Number | 3.1.1.104 |
| Gmelin Reference | 60705 |
| KEGG | C12233 |
| MeSH | D000072609 |
| PubChem CID | 6917953 |
| RTECS number | VR8400000 |
| UNII | 9V1DE99H0Z |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID7054267 |
| Properties | |
| Chemical formula | C11H16O2 |
| Molar mass | 222.34 g/mol |
| Appearance | White to light yellow crystalline powder |
| Odor | Odorless |
| Density | 1.009 g/cm3 |
| Solubility in water | Insoluble in water |
| log P | 3.78 |
| Vapor pressure | 0.0000678 mmHg at 25°C |
| Acidity (pKa) | 10.03 |
| Basicity (pKb) | 9.27 |
| Magnetic susceptibility (χ) | -70.0e-6 cm³/mol |
| Refractive index (nD) | 1.537 |
| Viscosity | Viscous liquid |
| Dipole moment | 3.37 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 427.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -6358.3 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | D11AX18 |
| Hazards | |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H315: Causes skin irritation. H319: Causes serious eye irritation. |
| Precautionary statements | Precautionary statements: "P261, P264, P271, P272, P273, P280, P302+P352, P305+P351+P338, P312, P321, P332+P313, P362+P364, P501 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 113°C |
| Lethal dose or concentration | LD50 (rat, oral): >2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 3,150 mg/kg (rat, oral) |
| NIOSH | DW0S225DJT |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.1% |
| Related compounds | |
| Related compounds |
Olivetol Cannabigerol Resorcinol Phloroglucinol Alkylresorcinols |