Chemical advances shape entire industries, and the emergence of 2,4-Xylenol reminds me how much one substance can change the direction of technology. Around the end of the nineteenth century, chemists started isolating xylenol isomers during coal tar distillation. The focus sharpened on 2,4-Xylenol by the early 1900s, when researchers recognized unique properties compared to its methylphenol relatives. By the mid-twentieth century, industrial use motivated large-scale synthetic pathways, moving away from extraction to more controlled manufacturing. Once its usefulness became clear, companies invested in new purification and characterization techniques, which led to far more reliable supplies for agriculture, chemical synthesis, and antiseptic applications. Decades of innovation turned what was once a lab curiosity into a mainstay for several sectors.
Folks familiar with industrial chemistry will recognize 2,4-Xylenol as a colorless to pale yellow crystalline solid, with a sharp, medicinal scent that hints at its roots in coal tar and cresylic acid mixtures. Commercial suppliers manufacture bulk volumes for everything from disinfectants to adhesives. Chemical suppliers often offer both technical and reagent grades, catering to workers who design resins for the plastics industry, chemists customizing antioxidants, and research teams searching for new herbicide precursors. In the lab, the crystalline powder stands out due to its distinct solubility, making it easy to separate and process, either as a feedstock or finished product.
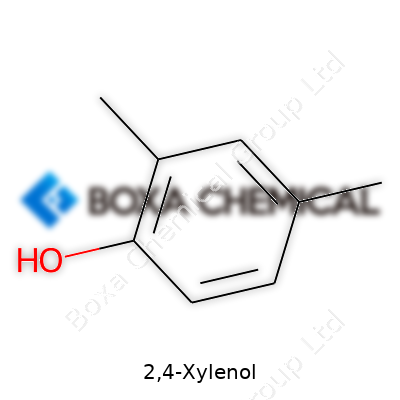
Examining this compound on the bench reveals quite a set of characteristics. 2,4-Xylenol, also called 2,4-dimethylphenol, has the formula C8H10O. At room temperature, it forms needle-like crystals or a granular powder with a noticeable aromatic odor. Its melting point falls around 45°C, allowing for easy melting and recrystallization—something a lot of chemists appreciate during purification. Boiling occurs near 210°C, and its vapor pressure is moderate, which simplifies handling in industrial settings but requires caution for large-scale venting. The compound dissolves in organic solvents like ethanol, ether, and benzene, but it only shows slight solubility in water. This characteristic limits some routes of environmental mobility but heightens awareness when considering safety protocols. Its phenolic hydroxyl group enables strong hydrogen bonding, leading to moderate acidity and reactivity in substitution reactions.
Chemical manufacturers provide various specifications to support customers from formulation to analytical testing. Labeling includes CAS number 105-67-9, purity percentage (frequently at least 99%), and a profile of potential residuals such as other xylenol isomers or cresols. Safety data sheets address hazard pictograms (corrosive and harmful labels), boiling point, melting range, density (around 0.965 g/cm³ at 20°C), and recommended maximum storage temperatures. Container labels must state batch number and manufacture date, and some also mention recommended handling methods in line with global regulatory standards, reflecting the chemist’s need for exact traceability. High-grade products come with detailed certificates of analysis, sharing spectroscopic data or chromatographic purity, especially when the compound is headed for research or specialty synthesis.
For a long while, obtaining 2,4-Xylenol meant fractionating coal tar, a byproduct of coking plants, using advanced distillation and extraction. Industrial labs soon developed synthetic methods, with the cumene process or methylation of phenol overtaking older approaches for reliability and efficiency. Alkylation, using methanol and an acid catalyst with phenol, rose to prominence due to better yields and controllable reaction conditions. The process produces a mix of xylenol isomers, but chemical engineers fine-tune conditions—temperature, pressure, catalysts—to optimize for the 2,4 isomer. Liquid-liquid extraction and recrystallization remain pivotal in isolating the desired product, offering a lesson in balancing scalability, environmental responsibility, and purity.
2,4-Xylenol’s chemical structure—two methyl groups flanking the phenolic ring—changes how the compound reacts compared to classic phenol. Reactivity centers on the aromatic ring and hydroxyl group, with alkyl and halogen substitutions ranking high among modification strategies. Nitration, sulfonation, and etherification all yield a versatile library of spin-off molecules, which helps fine-tune antioxidants or produce intermediates for rubber and polymer production. Oxidative coupling can form oligomers useful in specialty resins or adhesives. The electron-donating methyl groups boost nucleophilicity and shield certain positions, allowing better selectivity during synthesis. These reaction options make 2,4-Xylenol a useful building block for chemists eyeing innovations in advanced materials or pharmaceuticals.
Across catalogues and safety databases, you might spot 2,4-Xylenol under an array of synonyms: 1-Hydroxy-2,4-dimethylbenzene, 1,3-dimethyl-4-hydroxybenzene, or just 2,4-dimethylphenol. In the trade world, broad labels such as “dimethylphenol” sometimes cause confusion, especially for those new to chemical supply. Some brands market it as part of cresylic acid blends, factoring in other methylphenol isomers. Accuracy remains key—regulatory filings require correct naming, while cross-checking synonyms is non-negotiable to maintain safe and effective storage and transport.
Workplace safety matters a lot where 2,4-Xylenol appears on the inventory list. Inhaling dust or vapors leads to throat or lung irritation, and even brief skin contact often results in burning and redness. Extensive research confirms toxicity via oral, dermal, or inhalation exposure, and cases of high-dose poisoning have brought about strict regulation. Facilities handling this compound must supply eye protection, gloves made from solvent-resistant materials, and enclosed ventilation systems. Eye-wash stations become non-negotiable pieces of kit. Safe storage calls for sealed containers away from direct sunlight and sources of ignition. Any risk of spills turns the spotlight on training—workers need real drills, not just written instructions. Regulatory agencies worldwide—like OSHA, EU-REACH, and China’s SAWS—set exposure limits and reporting requirements, pushing producers to offer transparent, up-to-date safety data sheets.
2,4-Xylenol weaves into a surprising array of end products. Disinfectants and antiseptics tap its germ-killing power, bringing peace of mind to clinical and household environments. In synthetic chemistry, it kicks off production of high-performance resins, adhesives, and rubber chemicals. Manufacturers of electronic components use it as a stabilizer in plastics, where thermostability and resistance to degradation count for a lot. Agriculture draws on 2,4-Xylenol derivatives for certain herbicides. Even the fragrance sector explores its derivatives, though always with an eye on toxicity and regulatory guidance. Chemical engineers appreciate its flexibility, welcoming new uses that stem from active research and close collaboration with downstream industries.
Labs worldwide continue exploring fresh ways to modify and deploy 2,4-Xylenol, searching for molecules that pack more function into a smaller space. Polymer chemists trial new cross-linking agents to push boundaries of strength and flexibility. Medicine looks for innovative derivatives with higher selectivity as antimicrobial or antifungal agents. Process chemists experiment with green chemistry, seeking catalysts that trim energy use and waste streams. Spectroscopic and chromatographic advances offer sharper tools for ensuring purity and identifying unwanted by-products. Teams specializing in material science use 2,4-Xylenol-based copolymers for specialty coatings, striving for better corrosion protection and higher performance in harsh environments. Competitive companies fund research into sustainable feedstocks and circular manufacturing loops, signaling a future where waste drops and cost savings grow without compromising worker safety or environmental responsibility.
Interest in toxicity dates back over a century, and few compounds get as much scrutiny among methylphenols as 2,4-Xylenol. Ingesting, inhaling, or absorbing this substance carries acute risks—central nervous system effects, hemolytic anemia, and damage to kidneys and liver. Researchers map dose-response curves and chronic exposure outcomes, which enable accurate risk assessments for workers in chemical plants or emergency responders. Studies reveal that repeated low-level exposure, especially in unventilated spaces, increases risk for dermatitis and systemic effects, pressing the case for strict threshold limits and medical surveillance. Animal data point to dose-dependent toxicity, but ongoing research focuses on mechanisms at the cellular level, searching for ways to limit accidental harm and refine remediation strategies for contaminated sites. Regulatory agencies keep gathering fresh evidence, refining classification and broadening protective measures as new studies come in.
Chemical innovation thrives on compounds with versatility, and 2,4-Xylenol stands out for the range of possibilities still ahead. Performance polymers, advanced adhesives, and smart coatings use this compound as a starting point, giving designers something to build on for sectors as varied as electronics, clean water, and packaging. Growing attention toward green chemistry motivates companies to revisit old manufacturing pathways, trialing recyclable catalysts and renewably sourced feedstocks. Digital modeling tools make it easier to simulate new reactions and predict toxicological outcomes before real-world testing begins. As regulatory expectations climb and customers demand safer, more transparent ingredients, producers must keep refining practices. Workers in research, safety, and manufacturing play critical roles—by sharing data, updating best practices, and designing safer derivatives—so 2,4-Xylenol retains both its relevance and reputation for responsible use.
Everyday life depends on more chemicals than most folks realize. 2,4-Xylenol is one of those behind-the-scenes compounds, giving a quiet boost to health, industry, and cleanliness. Chemists call it a "phenolic compound," but to most people, it’s just part of the mix that keeps harmful germs in check or helps make sure other products work as advertised.
Walk into a hospital, open the cabinet, and check the labels on some antiseptic liquids or soaps. There's a good chance you’ll spot 2,4-Xylenol in the fine print. It plays a key role in fighting off bacteria and preventing infections. Most commonly found in disinfectants, 2,4-Xylenol sticks out for its ability to tear down cell walls of dangerous microbes. A quick wipe or scrub releases it to finish off bacteria before they multiply and cause bigger problems. It’s far from perfect—like any chemical, some bacteria learn to resist over time, but for now, it keeps environments a lot safer than they'd be without it.
The same properties that make it tough on germs have put it in paints, wood preservatives, and coatings. Mold, mildew, and rot like to tear into materials that get damp and dirty. 2,4-Xylenol thwarts this, stretching the life of wood, textiles, and paint jobs. Homes, schools, and public buildings use these treated products to avoid early replacement or expensive repairs.
Not every job for this compound revolves around fighting bugs. Chemical manufacturers rely on it to make other useful substances—think herbicides, dyes, and pharmaceuticals. It serves as a building block for all kinds of specialty chemicals modern society has come to count on. Without folks refining and reworking 2,4-Xylenol, the chain reaction to produce some lifesaving drugs and crop protection products would grind to a halt.
From time to time, I've spoken with people who have handled chemical production. Their main concern stays the same: safety. 2,4-Xylenol is powerful, but it needs respect. Mishandled, it causes irritation, and can trigger issues for workers' skin, eyes, and respiratory systems. Strong rules and good training make the difference between safe output and serious workplace accidents.
Each powerful ingredient has a double edge. If too much 2,4-Xylenol gets into the air, water, or soil, it can hurt wildlife and seep into drinking water. Studies from the US Environmental Protection Agency show that even small leaks add up in the long run, putting local animals and humans at risk. In regions where factories dump waste without proper treatment, nearby families have found water sources tainted and crops struggling.
Manufacturers have begun using better filters, scrubbers, and waste treatment solutions. Stricter government oversight has pressed companies to clean up their act—quite literally. Watchdogs and local communities push for regular inspections and independent monitoring, making it easier to catch and halt improper disposal before it spirals.
Anyone using products that contain or rely on 2,4-Xylenol counts on those making it to put safety and honesty above short-term profit. Alternatives exist for some uses, but not every replacement stands up to the test yet. So, responsible use, strong oversight, clear labeling, and ongoing research carry just as much weight as the compound itself.
2,4-Xylenol crops up in conversations about chemicals used for disinfectants, resins, and sometimes even in the creation of antioxidants. The stuff has a strong chemical odor and can be tricky to handle for those not wearing the right gear. Just because a compound pops up in manufacturing doesn’t make it safe for people or the planet. I’ve seen more than a few workplace safety posters that mention xylenols for a reason.
Spilling a bit on your skin usually ends with irritation — redness, itchiness, maybe even burns if you don’t wash up fast. My friend who works at a chemical plant told me stories about colleagues who rushed to the eyewash station after just a splash. People breathe in the vapors by mistake, and it hits their lungs hard: coughing, headaches, and sometimes trouble breathing for hours. Health experts, including folks at the CDC, put 2,4-xylenol on lists of substances that demand careful handling. That observation isn’t just a formality — it’s based on documented incidents.
Folks who work around this compound over the long haul encounter bigger risks. The science points to possible liver and kidney stress, and a few animal studies even link repeated exposure with changes to blood chemistry. Sure, the animal data doesn’t always match up with human reactions, but ignoring those results never pans out well. The International Agency for Research on Cancer keeps a close eye on compounds like xylenols but hasn't labeled 2,4-xylenol a cancer risk as of now. That said, data gaps shouldn’t give anyone a free pass to be careless.
Chemical runoffs have a way of finding their own path, sometimes sneaking into groundwater and affecting fish or plant life downstream. 2,4-Xylenol breaks down over time, but not before it disrupts aquatic life and soils. The U.S. Environmental Protection Agency keeps tabs on substances like this to keep drinking water and food sources clean. I used to live near a river that caught runoff from a factory: dead fish, funny smells, and thick patches of algae showed me that labeling something as “low-risk” in a lab doesn’t always match reality out in the world.
Reducing risk starts in the workplace. Anyone handling 2,4-xylenol should get gloves, splash goggles, and fans or exhaust hoods to pull away dangerous air. Training isn’t just paperwork — practical drills help workers respond fast if things go wrong. For companies, closed-loop systems limit how much vapor escapes and make spills less likely. Regular air monitoring can help spot a problem before it gets worse.
Outside of the plant, strict spill reporting and clean-up rules make a big difference. Following EPA waste disposal rules cuts down on environmental impact. Research into different compounds for the same job might someday sideline 2,4-xylenol, lowering workplace dangers and helping out the environment.
Anyone involved with 2,4-xylenol — from manufacturers to workers to neighbors — should keep an open line with local health officials and regulatory agencies. Looking at real-world data, listening to medical advice, and supporting new research offers a smarter way forward than taking comfort in routine. As workers and members of a wider community, we only benefit from clear-eyed risk awareness and strong protections.
Most folks underestimate chemical storage until something goes sideways. In my early years on the shop floor, one poorly placed drum of solvent taught the crew what “too late” smells like. 2,4-Xylenol has a similar bite. It brings risks you don't want in tight corners, so treating storage with the respect it earns is practical safety, not just regulation.
2,4-Xylenol tends to form crystals at lower temperatures and carries flammable label warnings. According to the National Fire Protection Association, it sits in fire hazard category 2 because its flash point comes in under 93°C. Even a seemingly small slip in temperature control can push it toward solid form or, worse, fumes. That alone pushed my own process team—and plenty of my peers—to invest in reliable thermometers and alarms.
It makes sense to stash this compound in cool, dry places, away from direct sunlight and sources of heat. Humidity and heat push up vapor pressure and, with flammables, this means a real fire hazard. Keeping it in a sealed, properly labeled container keeps evaporation at bay and keeps workers from guessing what's inside. OSHA stresses original containers for a reason: relabeling or splitting into other bottles opens the door to handling errors. Personal experience taught me that unlabeled containers invite disaster.
Never ignore air flow around storage—adequate ventilation means vapors don’t collect and linger. Old warehouses with stagnant air let fumes build up after a leak, creating risks to workers and even folks in nearby offices. I once toured a plant that had to shut down for hours just to vent a pocket of fumes from poor storage design. Investing in mechanical ventilation—like exhaust fans and properly placed vents—cuts that risk. Using explosion-proof equipment removes another worry.
2,4-Xylenol should not bunk up with oxidizers or acids. Many accidents happen not because of what’s being stored, but because of what’s stacked beside it. Mixing flammables with incompatible chemicals ranks high on OSHA’s most cited violations. Separate storage areas—marked by clear signage—make sense in every facility, no matter the scale. Even in smaller workshops, that lesson is king.
In my time with safety audits, I learned that personal protection starts with smart layout. Workers want gloves, goggles, and respirators handy when handling or cleaning up after spills. In one warehouse visit, a missing chemical shower turned a small spill into a big medical bill. No one expects a splash; PPE needs to be easy to reach and always stocked.
Routine checks of all containers, including seals and correct labeling, cut down on both big and small incidents. No safety gear can outwork basic vigilance and regular audits. Spills always happen on the day everyone assumes nothing will.
Solid documentation tracks inventory, movement, and storage conditions, keeping small errors from snowballing. Training turns regulations into habits. In shops where every worker knows the chemical storage rules, accidents and near misses drop off a cliff. Employers make life easier (and safer) by posting safety data sheets in plain sight and reviewing procedures with every shift.
Handling 2,4-Xylenol in a responsible way sets the tone for the whole operation. Smart processes, updated training, and a respect for every drum or bottle on the shelf keep people healthy and businesses running.
Step onto any site where 2,4-Xylenol shows up—factories blending resins, labs mixing antioxidants, facilities running specialty chemical production—and the same warnings pop up. This stuff does a lot for industry. It's tough and reliable, powering everything from adhesives to disinfectants. It’s also stubbornly hazardous. Too many workers have felt its sting, not just on paper but in real life, where a careless whiff or a splash leaves burns, dizziness, or worse.
Handling this compound has shaped my respect for chemistry. Its strong smell hangs in the air long after a spill. Without gloves or goggles, skin starts to itch or burn. The vapor sneaks up, and even a quick taste in the throat leaves workers coughing or wheezing. The Centers for Disease Control and Prevention lists 2,4-Xylenol as a potential irritant to skin, eyes, and the respiratory system. Long-term exposure can mess with the liver and kidneys. Once, at a production site, a maintenance worker ignored chemical-resistant gloves for “just a second”—he spent four hours in the ER because xylenol won the bet. It’s not just about scaring people; these real-world cases say more than warning labels.
Safety isn’t about locking down a lab. It means looking after coworkers and heading home healthy every day. Simple habits make the most impact. Nitrile gloves keep the liquid off your hands. Splash-proof goggles save eyes from accidental sprays. Not all dust masks block the vapor—approved respirators with organic vapor cartridges do a better job in closed spaces. Thick aprons and proper shoes really reduce accidental contact. Clean, working gear stands between a normal workday and a bad accident.
Chemical storage keeps 2,4-Xylenol away from open flames, oxidizers, and acids. Good ventilation and hoods cut down on dangerous fumes. Designated chemical storage rooms with real labeling—no faded signs—let everyone know what’s inside. Training isn’t wasted time. It covers how to lift, how to pour, how to mop up without spreading a spill. The Occupational Safety and Health Administration spells out regulations that work, but no federal rule matches experience gained from real training. Ask a technician who’s cleaned up just one improper spill; the lessons stick.
Protocols work best when folks trust them. If anyone feels uncomfortable with a process, respect that gut feeling. Open reporting helps spot near-misses and small leaks before they become ambulance calls. Keep emergency showers and eyewash stations clear and tested. Inventory logs, inspection checklists, and regular team drills build responsible routines. After plenty of years in environments with dangerous chemicals, I’ve seen teams who laugh off protocols. Those places rack up injuries. Teams who take ten seconds to double-check stay on the right side of health.
No chemical makes shortcuts worth the risk—not this one, not any. 2,4-Xylenol rewards those who work with steady hands and clear heads. If people look out for each other and never treat safety like a chore, workplaces become both productive and trustworthy. Safe handling of hazardous chemicals isn’t theory; it’s the lived truth for anyone who earns their living in the chemical world.
2,4-Xylenol has shaped more than a few memories in my time spent wrangling jars in the lab. The stuff packs a heavy, sharp odor—think of riding past a fresh strip of tar on a hot road. Truth is, that scent comes from a chemical packed with physical quirks that matter to people working in industries that value both muscle and precision.
Grab a small bottle of 2,4-xylenol at room temperature, you’ll see a pale, yellowish crystal or something that could pass for a thick, stubborn liquid when it gets a little warmth. Its melting point lands around 26 degrees Celsius, which lines up with that halfway-between-solid-and-melted texture you’ll catch on a warm day. It melts quickly and that makes a difference when manufacturers need ingredients to blend fast, not clump.
This compound holds enough weight for easy handling (about 122 grams per mole), but what always catches my attention is its stubborn refusal to dissolve in water. Throw a scoop into a beaker hoping for a quick mix? All you’ll get is a mess clumping to the sides. Toss it in a bottle with organic solvents like alcohol or ether, and you’ll watch it blend smoothly without much resistance.
Every functional chemist knows that the two methyl groups parked on the benzene ring of 2,4-xylenol and that lone hydroxyl group set the stage for reactivity. These positions (the “2,4” in its name) put this molecule squarely in the bracket of phenols—famous for their antiseptic bite and ability to kill germs. You get a sense of the real-world impact in disinfectants or wood preservatives where this property pulls a lot of weight.
I’ve seen its moderate acidity come in handy during tests—its pKa sits at about 10, so compared to everyday carboxylic acids, it won't give up its hydrogen lightly. But ask it to fight off a weak base and it will release a hydrogen ion quickly enough to change the outcome of a reaction. In practice, that means it plays well when neutralization reactions are essential for product safety.
Looking past the microscope, these details mean something in real terms. Industries rely on the low melting point to manufacture substances ranging from resins to surface coatings. Its insolubility in water helps ensure chemical formulations stay where they’re needed, so products perform as promised without unforeseen breakdowns. Its reactive nature opens the doors to antiseptics, antioxidants, and stabilizers.
Chemicals aren’t just theoretical puzzles—they land in our homes, hospitals, and farmlands. Misuse, though, can mean risks. 2,4-Xylenol’s toxicity and strong aroma warn the user to keep safety goggles and gloves handy, and remind us why automated ventilation matters in labs and factories. The answer isn’t to shy away from these risks but to train workers, enforce labeling, and keep emergency protocols current.
It’s not every day I run across a material as stubborn but useful as 2,4-xylenol. To keep folks safe and products effective, industry watchdogs have ramped up guidelines on storage, from fire-resistant containers in labs to ventilated spaces for larger stockpiles. Advances in personal protective equipment—masks, gloves, and full suits—give another layer of certainty. On the chemistry side, new research aims to turn waste streams into less hazardous byproducts, stretching both resourcefulness and responsibility further than before.
So working with 2,4-xylenol isn’t about leaning on habit or convenience. It’s a tug-of-war between extracting its value and steering away from danger. Experience and respect for its properties mark the difference between routine and disaster—a lesson written in every strong aroma and stubborn crystal that crosses my bench.
| Names | |
| Preferred IUPAC name | 2,4-dimethylphenol |
| Other names |
B-(2,4-dimethylphenol) 2,4-Dimethylphenol 2,4-Xylenol 2,4-Xyleneol 4-Hydroxy-m-xylylene |
| Pronunciation | /tuː fɔːr zaɪˈliːnɒl/ |
| Identifiers | |
| CAS Number | 105-67-9 |
| Beilstein Reference | 2041191 |
| ChEBI | CHEBI:17338 |
| ChEMBL | CHEMBL1549 |
| ChemSpider | 1247 |
| DrugBank | DB11360 |
| ECHA InfoCard | 03b1a713-2e09-4aad-bf58-6e7dbb88bd14 |
| EC Number | 2.4.2.23 |
| Gmelin Reference | 82294 |
| KEGG | C01418 |
| MeSH | D015801 |
| PubChem CID | 13615 |
| RTECS number | ZE2450000 |
| UNII | F97MCS535J |
| UN number | UN2229 |
| Properties | |
| Chemical formula | C8H10O |
| Molar mass | 122.16 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | phenolic |
| Density | 1.02 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.23 |
| Vapor pressure | 0.24 mmHg (at 25 °C) |
| Acidity (pKa) | 10.33 |
| Basicity (pKb) | 10.10 |
| Magnetic susceptibility (χ) | -64.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.531 |
| Viscosity | 2.78 mPa·s (at 25 °C) |
| Dipole moment | 1.17 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 137.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -48.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4035.6 kJ/mol |
| Pharmacology | |
| ATC code | D08AE02 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation, toxic to aquatic life. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H302, H315, H318, H335 |
| Precautionary statements | P264, P280, P305+P351+P338, P310, P501 |
| NFPA 704 (fire diamond) | 3-2-0 |
| Flash point | 79 °C (174 °F; 352 K) |
| Autoignition temperature | 530°C |
| Explosive limits | 1.3-9.8% |
| Lethal dose or concentration | LD50 oral rat 1210 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1210 mg/kg (oral, rat) |
| NIOSH | SA0875000 |
| PEL (Permissible) | 50 ppm |
| REL (Recommended) | 15 mg/m³ |
| IDLH (Immediate danger) | IDLH: 50 ppm |
| Related compounds | |
| Related compounds |
Cresol Phenol Resorcinol BHT (Butylated Hydroxytoluene) Thymol |