Long before 2,3-Xylenol showed up in chemical catalogs, phenolic compounds attracted the attention of early researchers hunting for useful derivatives from coal tar and crude oil distillates. Chemical pioneers focused on separating the various isomers of xylene and their hydroxylated cousins once they noticed these molecules held both industrial promise and environmental challenge. Over decades, processes evolved and synthetic methods improved, making what once was a tiny curiosity into a widely studied organic intermediate.
2,3-Xylenol, sometimes found labeled as 2,3-dimethylphenol, serves as an important intermediate in both laboratory and industrial settings. You will notice clear, pale yellow crystals or oily liquids depending on the batch and temperature, and a very distinct phenolic scent that signals its presence long before a label does. Bulk suppliers and specialty chemical producers alike keep a close eye on purity and impurity profiles for this compound, since even small amounts of byproducts can impact downstream applications.
This aromatic phenol carries the molecular formula C8H10O, or more specifically, C6H3(CH3)2OH at the 2,3-positions, with a molecular weight right around 122.17 g/mol. Look for melting points between 42°C and 45°C, with boiling coming in close to 210°C. The compound dissolves in alcohol and ether but shows limited solubility in water. Its chemical reactivity centers on the phenolic OH group, while the two ortho-positioned methyl groups add steric hindrance and electron donation, which tends to push its oxidation and substitution reactivity in different directions compared to its 2,4- or 3,5-isomers.
Industry standards for 2,3-Xylenol get set by both chemical purity and water content, along with a tight limit on sulfur, halide, and metallic impurities. Most commercial documentation provides batch numbers, CAS Registry Number (526-75-0), UN Hazard Code (if shipping internationally), and manufacturing or expiration dates. Safety data sheets, strictly enforced in labs and warehouses, require freshly updated toxicological and handling data, because older MSDS documents may miss new regulatory controls or findings from the latest animal model research.
Early chemists isolated xylenols through exhaustive distillation from wood tar and coal tar fractions, followed by laborious crystal separation. Today, oxidative methylation routes, often catalyzed by copper or vanadium systems, dominate high-volume production cycles. One typical industrial method starts from o-xylene, with methylation and phenolation steps under strictly controlled conditions to favor the 2,3 arrangement over the less valuable isomers. Each batch gets refined using vacuum distillation and liquid-liquid extraction, then passes through charcoal beds to strip out colored impurities before final crystallization and packaging.
An experienced synthetic chemist can coax 2,3-Xylenol into a wide range of downstream products. Halogenation, alkylation, sulfonation, and nitrosation provide favored pathways for custom intermediates. The methyl groups can block or direct new substituents, protecting certain bonds during multi-step synthesis, making this isomer especially useful for building block strategies in pharmaceutical and agrochemical research. During oxidation, the compound yields dialdehydes or acids, and through etherification or esterification, it’s possible to anchor xylenol into polymer backbones or surfactant chains. In the laboratory, researchers often turn to 2,3-Xylenol for chelating ligands, UV stabilizers, and even enzyme mimetics, thanks to its tuning potential.
An order for 2,3-Xylenol might also show up under its less formal names: 2,3-dimethylphenol, o,m-xylenol, or UNII code S8N5DJG30V. Some boutique suppliers label it as methylphenol 2,3-isomer or “mixed xylenol, ortho/meta rich.” Tracking synonyms makes life easier for compliance teams and reduces mislabeling headaches, especially with import/export controls that cross-check UN numbers and harmonize names against big chemical inventories.
Lab safety officers and industrial hygiene experts keep a close watch on 2,3-Xylenol handling practices. Direct contact can irritate eyes, skin, and respiratory system; inhaling the fumes brings risk of dizziness or even systemic toxicity. Typical protocols involve nitrile gloves, fume hoods, and sealed containers that minimize vapor escape. Storage guidelines steer clear of high heat and strong oxidizers, and spill kits must include absorptive material rated for phenolics. Facilities carrying out scale-up or pilot plant runs will frequently install continuous monitoring for airborne concentrations and backup ventilation in case fans quit during critical operations. Regulatory agencies set workplace exposure limits and mandate up-to-date training for workers who handle the material more than once in a blue moon.
As a high-value intermediate, 2,3-Xylenol finds its way into far more than academic labs. Its core structure becomes the starting block in manufacturing fungicides, photographic developers, specialty dyes, and high-performance resins found in electronics or coatings. Over the years, its role expanded into the synthesis of custom antioxidants, flavor agents, and polymer additives that shore up the durability or thermal properties of end products. Research groups studying enzyme functions use substituted xylenol analogs to mimic phenolic oxidations in biological systems. Demand ebbs and flows with market needs for downstream specialties, forming a reliable, if niche, pillar of the specialty organics industry.
Active R&D efforts have focused on new production routes that slash energy use, lower greenhouse gas emissions, or recycle previously wasted co-products from related xylene processing. Analytical chemists have published refinements to GC-MS methods for trace detection, raising hopes for smarter process control and lower loss rates. Some research teams keep tuning substituents around the phenol ring to design molecules with target selectivity as anti-corrosives, drug precursors, or even biomedical probes. The flexibility of the compound’s core means patents keep stacking up for ways to turn minor changes around the ring into real-world advantages, especially in catalysis and electronics.
Few topics bring more scrutiny than the health risks of small-molecule organics. Rodent studies show 2,3-Xylenol produces dose-dependent effects spanning mild irritation to systemic organ toxicity. In aquatic settings, the compound threatens sensitive species, urging strict effluent controls from facilities processing phenols. Researchers probing metabolic breakdown have discovered both glucuronide and sulfate conjugates in mammalian systems, helping guide regulations on permissible workplace levels. Occupational health teams focus on short- and medium-term exposure, collecting blood and urine samples to correlate any chronic low-dose effects. Regulators respond with evolving workplace safety laws pushed by new data and monitoring technologies.
Development prospects for 2,3-Xylenol look tied to broader shifts in green chemistry and materials science. Producers watch for breakthroughs in catalysis that lower the carbon footprint of synthesis and recovery. As electronics miniaturize, demand for more stable, heat-resistant polyphenols will likely drive innovation in derivative manufacturing. Environmental teams target new degradation technologies to break down xylenols before they reach local water systems. Risk teams want predictive modeling that links structure to acute or chronic toxicity, using computational chemistry to flag hazards early in development. In all of this, 2,3-Xylenol stands less as a common commodity and more as a specialized, versatile building block—one whose story marks the ongoing intersection of chemistry, industry, and public health.
2,3-Xylenol may not sound like something you encounter every day, but it shows up in more places than most people realize. This compound, part of the xylenol family, brings value for both its chemical structure and how it behaves in the real world. For those of us who take for granted things like disinfectants or resins used in electronics, this is the sort of ingredient quietly doing heavy lifting.
Looking at practical uses, 2,3-Xylenol finds a spot in disinfectants. From my own experience dealing with cleaning supplies in a small auto shop, labels often list “phenolic compounds,” and 2,3-Xylenol is one of them. These substances prove useful because they break down bacteria and prevent mold – something that matters a lot where oil, grease, and moisture invite stubborn messes. Hospitals and clinics also lean on compounds like these for sanitizing surfaces, especially because they hold up under harsh conditions and keep working even after repeated cleaning cycles.
Factories making resins and plastics use 2,3-Xylenol as a building block. It’s a stepping stone in the production of tougher, more stable materials. Every time I mess with a circuit board while fixing old gadgets, I’m reminded that the thin films and epoxy layers owe their properties to chemicals like this. The electronics industry leans on these materials since they don’t melt or warp under heat and keep out moisture, both critical to making reliable devices. There’s a confidence that comes from knowing these resins protect expensive hardware and keep everything running, from power tools to medical equipment.
2,3-Xylenol also crops up as a starting material for dyes and pigments. Textile and paint makers depend on reliable colorants to produce vibrant, lasting colors. I’ve seen the difference quality pigments make: cheaper paints fade fast, and fabrics lose their appeal. Stability, color strength, and resistance to sunlight become major selling points, and the chemistry behind the scenes ensures that products don’t disappoint.
In the pharma world, chemists use 2,3-Xylenol in synthesizing drugs. Some painkillers, antiseptics, and intermediates for more complicated medicines count on this compound at some stage. Farmers and agronomists benefit as well, since it’s involved in creating intermediates for pesticides and herbicides. This link to agriculture matters because crop yield and food safety face threats from pests in every region.
Like a lot of chemicals, 2,3-Xylenol won’t win awards for environmental friendliness without careful handling. It can be toxic if mismanaged or dumped without treatment. I’ve read stories about soil and water contamination forcing entire communities to rethink their industrial policies. Regulations exist, but enforcement varies, and risk often comes down to how scrupulous factories remain in their operations.
It doesn’t take much imagination to see the solution: stricter oversight, support for greener alternatives, and investments in safer production. Some companies invest in recycling waste streams and developing less toxic substitutes, and that kind of effort pays back in trust and long-term sustainability. For anyone who cares about safer consumer goods and a healthier world, taking a hard look at chemicals like 2,3-Xylenol seems like a smart bet.
2,3-Xylenol pops up in more places than most people realize. Used in factories, labs, and even as a starting block for making other chemicals, this substance often lurks behind the scenes in paint thinners, disinfectants, and some plastics. It doesn’t take much to appreciate why looking at its health risks matters. Day-to-day exposure in jobs, accidents in transport or storage, or just working close to certain industrial processes brings this tough question: what does 2,3-xylenol do to people and the environment?
Chemists describe 2,3-xylenol as both flammable and, in enough quantity, toxic. What does this mean for workers, bystanders, or the person who has to clean up a spill? Breathing vapors can sting the eyes, irritate the nose, trigger coughing, and leave people feeling light-headed or nauseous. Touching it for too long without gloves sometimes causes skin rashes or burns. Reports of headaches, dizziness, or an upset stomach among exposed workers aren't rare.
Animal tests point to deeper trouble above a certain dose. The National Institute for Occupational Safety and Health highlights that, in high enough amounts, the compound harms the liver, kidneys, and central nervous system. It has not earned the label of a confirmed cancer risk, but any solvent that stresses the body’s detox organs deserves respect in handling. These signs line up with stories from chemical workers over decades. Small mistakes or careless practices raise the risk quickly.
I’ve seen how easy it is to overlook these sorts of hazards. New workers might skip gloves or masks for short tasks, thinking a little won’t hurt. Supervisors sometimes say, “It’s just a quick job.” But in jobs I’ve worked, problems rarely come from a single big mistake—small exposures, over days and months, add up. Health issues often sneak in quietly: mild headaches more often at the end of shifts, or unexpected skin issues after a busy season.
Cleanup teams have told me about challenges after tank leaks, where emergency crews faced hours of headaches or nausea. Public health records show that these incidents, while rare, bring long investigations and lasting concerns for people living nearby. Industry regulations, like those in OSHA standards, aren’t there for show—they’re built from incidents and sometimes tragedy. No shortcut or hasty fix ever makes up for a worker’s lost health.
The good news: most risks from 2,3-xylenol don’t need to spell disaster. Strong ventilation, proper personal protective equipment, and well-written emergency plans go a long way. Regular training, not just occasional reminders, keeps people alert. Shifting away from open containers and keeping hands away from the face cuts the chance of contact.
Disposal matters too. Tossing chemicals into regular trash or drains sends toxins downstream, filling up landfills with hazardous leftovers or polluting water. Waste treatment and storage, as outlined by the EPA, make a difference for the community.
The lesson here taps into a basic truth: what gets used or ignored inside a workplace ends up touching people and the planet. 2,3-xylenol isn’t the world’s worst toxin, but underestimating it often leads to avoidable harm. Companies, workers, and communities all have a stake in smart handling and clear information. Nothing about safety or common sense gets old. It’s one chemical story among many, and paying attention writes better endings for everyone involved.
2,3-Xylenol flows as a clear, colorless to pale yellow liquid, but don’t be fooled: this chemical can turn into real trouble if left unattended or handled like any run-of-the-mill solvent. Its sharp, pungent odor signals more than just inconvenience—it’s a red flag for toxicity. Vapors from 2,3-Xylenol cause eye, skin, and respiratory irritation. Too much exposure brings on headaches, nausea, and, in worst cases, central nervous system effects. Workers and anyone around this stuff can’t just pop open a window and let things slide.
The ideal spot for 2,3-Xylenol sits out of the sun, where cool temperatures fight off evaporation. Store containers in a dry place to keep moisture far away. Water invites corrosion, which sends the chemical leaking, and that’s often how workplace incidents start. Good ventilation isn’t just a buzzword. Every chemical supply room must keep air moving to push fumes out and away from unsuspecting lungs. Some warehouses have learned this the hard way when poor airflow left people woozy and sick.
Temperature swings spell danger, too. A hot warehouse means higher vapor pressure; more chemical vapor escapes and fills the air. Many industrial facilities use dedicated, temperature-controlled rooms for volatile organics. This approach has results—lower risks, fewer insurance claims, and fewer days lost to sick leave.
2,3-Xylenol catches fire more easily than many expect. Its flash point hovers around 87°C (188°F), so open flames, sparks, or even faulty wiring can start a fire. Static electricity isn’t just a lab theory either—it’s a real threat. Grounding and bonding containers, especially when transferring the chemical, prevent dangerous static charges from building up. Every safety manager knows stories of near-misses, all traced back to something as simple as an ungrounded drum pump.
Storing 2,3-Xylenol in steel or plastic drums with tight-fitting, chemically resistant lids keeps leaks and evaporation at bay. Improper containers put workers and operations at risk. Aging seals, cracked plastic, or corroded metal spell trouble. Regular inspections catch these problems before they turn into spills.
Mark all containers clearly with hazard warnings. In busy warehouses, clear labeling saves lives. Mixing up drums—mistaking a solvent for water—has led to real-world injuries. Color-coded systems and well-trained staff prevent those costly mistakes.
Solid training matters more than any fancy equipment. Every worker should know what they’re handling, what the hazards look and smell like, and exactly where to find spill kits and fire extinguishers. Annual drills make sure nobody forgets in a pinch. Some companies pair this with digital checklists and regular safety huddles—steps that pay off during OSHA visits and emergencies alike.
If a spill hits the floor, absorbents like clay or vermiculite soak up the mess. Never let spilled chemical go down the drain; catch it, bag it, and treat it as hazardous waste. Disposal rules vary, but every regulator agrees—2,3-Xylenol belongs nowhere near a storm sewer or dumpster.
Over the years, I’ve seen shops transform after one too many close calls. Better storage racks, improved air exchangers, thoughtful training—each change comes from recognizing risks, not from just checking chemical lists. Companies that take storage and handling seriously don’t just protect themselves from fines; they protect their workers, their neighborhoods, and their bottom line.
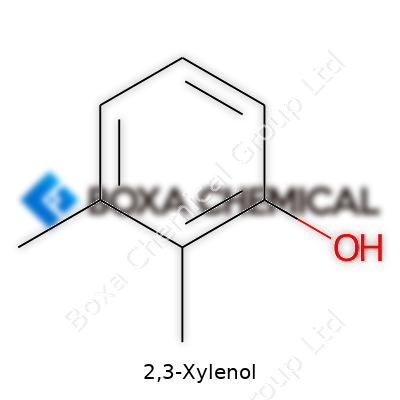
2,3-Xylenol stands out in the world of organic chemistry because of its arrangement on the benzene ring. It’s a methyl-dimethylphenol, which means you’ll see two methyl groups and a hydroxyl group attached to a classic six-carbon ring. The chemical formula is C8H10O. But numbers only tell part of the story. Placement matters, and here, the methyl groups sit on carbons 2 and 3, while the OH group claims the first spot.
During a university lab, the instructor brought out a sample and asked us for its structure. At first glance, it looked like another xylenol, but the slight shift in those methyl groups told a whole different tale. This taught me that small changes on a ring can completely change behavior and risk profile. Many xylenol isomers smell sharp and have strong disinfectant qualities, but shift a group, and you can find changes in boiling point and solubility.
Chemical plants see 2,3-xylenol as more than a laboratory curiosity. It turns up as an intermediate in dye production and serves the specialty resin markets. You may not recognize the name on a product label at the hardware store, but derivatives appear in adhesives and coatings—helping pigments last on surfaces and boosting durability in industrial paints. Engineers prize it because it mixes well in some solvents and interacts predictably in reactions critical for polymers.
More people today want transparency about chemical safety. Both the EPA and OSHA regulate phenolic compounds like this—exposure can irritate skin, eyes, or affect breathing. The Material Safety Data Sheet (MSDS) for 2,3-xylenol urges gloves and proper ventilation. I’ve seen careless handling in workshops end with a mad scramble for the eyewash station. There is a noticeable, unpleasant odor, a warning that your lungs and membranes deserve better protection.
Personal experience taught me not to skip the safety glasses. Never underestimate these volatile substances, even on a “quick test.” Over time, heavy releases into the environment could stress aquatic life, because phenolic compounds don’t break down fast in water or soil. That’s one reason companies invest in strict waste capture and recycling processes.
Researchers in green chemistry keep hunting for alternatives that break down faster or create less environmental impact. Some have shifted toward less toxic intermediates for resins and dyes, but the performance bar remains high. Regulations push for better capture of emissions and more effective worker protection. People inside and outside the field want less exposure risk and a smaller footprint.
Anyone working with substances like 2,3-xylenol gains a different kind of respect for what sits behind a product label. Knowing the chemical formula goes beyond memorization—it encourages questions about where it came from, where it ends up, and how it affects health over the long run. These aren’t just numbers—they guide choices in manufacturing, research, and safety culture.
Dealing with chemicals like 2,3-Xylenol takes patience, care, and a little know-how. This is not just any bathroom cleaner or garage oil; it’s a phenolic compound, showing up as a pale liquid with a strong smell, and its effects reach further than most realize. Tossing it down the drain can put a water system at risk, poison aquatic life, and spark environmental headaches many towns struggle to clean up.
A lot of people haven’t heard much about 2,3-Xylenol. Yet, it finds its way into research labs, chemical factories, and some specialty industries. The risk doesn’t end at direct contact—a simple spill seeps into soil, sneaks into groundwater, or gives off fumes that no one wants to inhale. The Centers for Disease Control (CDC) and the Environmental Protection Agency (EPA) both highlight its harmful effects on health, including irritation to skin, eyes, and lungs, and possible toxic symptoms after long exposure. Once it enters the ecosystem, nature can’t easily break it down.
Handling leftovers or outdated batches calls for a plan. Locking it away or ignoring it only delays a bigger problem down the road. Years in the lab taught me that trusted partnerships with licensed hazardous waste handlers matter more than anything else. Most universities and companies work with trained disposal services. These teams collect, label, and transport chemicals for treatment or incineration, following EPA and state rules.
Don’t ever try to neutralize 2,3-Xylenol with household chemicals or pour it in a dumpster. Mixed with acids, bases, or oxidizers, it reacts violently. Some stories show workers badly hurt after careless disposal—even small mistakes can lead to disaster. Everyone in the building shares the air and water supply, so responsibility stretches beyond the lab tech or supervisor on duty.
Sticking with closed, labeled containers keeps fumes at bay. Storing containers away from heat, light, and possible leaks limits fire and leak risks. Material Safety Data Sheets (MSDS) spell out specific risks and best practices, and every trained staffer reads those files before even opening a bottle. Many places now use barcodes to track inventory and trigger reminders when supplies near their end-of-life date.
For small businesses, community hazardous waste programs offer drop-off days or pick-up options. These services cost less than dealing with the aftermath of an accident or environmental fine. Local governments tend to list these programs online, and most respond quickly to help businesses or private citizens schedule safe pick-ups.
Some people think safe disposal rules just add red tape, but the benefits reach farther. Teaching employees and students to respect chemical waste keeps everyone safer. My earliest mentors didn’t just recite instructions—they shared real stories about what went wrong for those who cut corners. After seeing coworkers rushed to the hospital, few forget those lessons.
Innovation pushes toward greener chemistry, finding replacements for harsh substances like 2,3-Xylenol. Until then, following proven practices, supporting workers with proper training, and working with trusted disposal services give communities their best shot at staying healthy. Chemical waste may seem like someone else’s problem, but clean water and safe air connect every neighbor on the street.
| Names | |
| Preferred IUPAC name | 2,3-dimethylphenol |
| Other names |
2,3-Dimethylphenol 2,3-Xyleneol |
| Pronunciation | /tuː θriː zaɪˈliːnɒl/ |
| Identifiers | |
| CAS Number | 526-75-0 |
| Beilstein Reference | 604422 |
| ChEBI | CHEBI:16045 |
| ChEMBL | CHEMBL15714 |
| ChemSpider | 6407 |
| DrugBank | DB04219 |
| ECHA InfoCard | 100.010.341 |
| EC Number | 200-756-7 |
| Gmelin Reference | 62248 |
| KEGG | C01476 |
| MeSH | D015555 |
| PubChem CID | 13611 |
| RTECS number | ZE2625000 |
| UNII | 7T8FEC6H6O |
| UN number | UN2222 |
| CompTox Dashboard (EPA) | DTXSID9020700 |
| Properties | |
| Chemical formula | C8H10O |
| Molar mass | 122.16 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | phenolic |
| Density | 0.98 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.28 |
| Vapor pressure | 1 mmHg (20°C) |
| Acidity (pKa) | 10.18 |
| Basicity (pKb) | 9.86 |
| Magnetic susceptibility (χ) | -77.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.528 |
| Viscosity | 2.22 mPa·s (25 °C) |
| Dipole moment | 1.15 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 128.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -49.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3667.2 kJ/mol |
| Pharmacology | |
| ATC code | D02AE02 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H302, H312, H315, H318, H332, H335, H412 |
| Precautionary statements | P261, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 3-2-0 |
| Flash point | 86°C (187°F) |
| Autoignition temperature | 530 °C |
| Explosive limits | 1.3–7% |
| Lethal dose or concentration | 2,3-Xylenol: Oral rat LD50 = 1230 mg/kg |
| LD50 (median dose) | Rat oral LD50: 1210 mg/kg |
| NIOSH | UY3500000 |
| PEL (Permissible) | PEL: 5 ppm |
| REL (Recommended) | 75 mg/m³ |
| IDLH (Immediate danger) | IDLH: 50 ppm |
| Related compounds | |
| Related compounds |
Phenol Cresols 3,4-Xylenol 2,5-Xylenol |